  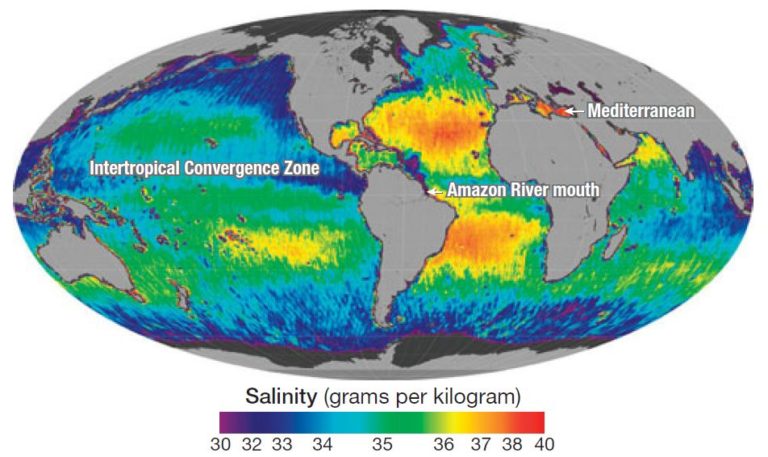
Its units are mS/cm (milli-Siemens per centimetre). salinity) and the mobility of the ions (ie temperature and pressure). The conductivity of sea water depends on the number of dissolved ions per volume (i.e. Again, minute differences occur between the old definitions and the new Practical Salinity Scale, but they are usually negligible. As the practical salinity is a ratio and therefore does not have units, the unit "psu" is rather meaningless and strongly discouraged. Some oceanographers cannot get used to numbers without units for salinity and write "35 psu", where psu is meant to stand for " practical salinity unit". Note that in this definition, salinity is a ratio and ( o/ oo) is therefore no longer used, but an old value of 35 o/ oo corresponds to a value of 35 in the practical salinity. The K value exactly equal to one corresponds, by definition, to a practical salinity equal to 35." The corresponding formula is: " The practical salinity, symbol S, of a sample of sea water, is defined in terms of the ratio K of the electrical conductivity of a sea water sample of 15☌ and the pressure of one standard atmosphere, to that of a potassium chloride (KCl) solution, in which the mass fraction of KCl is 0.0324356, at the same temperature and pressure. Since 1978, the " Practical Salinity Scale" defines salinity in terms of a conductivity ratio: The definition of salinity was reviewed again when techniques to determine salinity from measurements of conductivity, temperature and pressure were developed. The definitions of 19 give identical results at a salinity of 35 o/ oo and do not differ significantly for most applications. The United Nations Scientific, Education and Cultural Organization (UNESCO) decided to repeat the base determination of the relation between chlorinity and salinity and introduced a new definition, known as absolute salinity , It indicates a problem in the water samples used for the laboratory measurements. The fact that the equation of 1902 gives a salinity of 0.03 o/ oo for zero chlorinity is a cause for concern. The symbol o/ oo stands for "parts per thousand" or "per ml" a salt content of 3.5% 0 is equivalent to 35 o/ oo, or 35 grams of salt per kilogram of sea water. The relationship between salinity and chloride was determined through a series of fundamental laboratory measurements based on sea water samples from all regions of the world ocean and was given as Salinity was defined in 1902 as the total amount in grams of dissolved substances contained in one kilogram of sea water if all carbonates are converted into oxides, all bromides and iodides into chlorides, and all organic substances oxidized. The definition reflects the chemical titration process for the determination of chloride content and is still of importance when dealing with historical data. Chloride content was defined in 1902 as the total amount in grams of chlorine ions contained in one kilogram of sea water if all the halogens are replaced by chlorides. It allows determination of salt content through the measurement of a substitution quantity and calculation of the total of all material making up the salinity from that measurement.ĭetermination of salinity could thus be made through its most important component, chloride. The observation that - no matter how much salt is in the sea the various components present in a fixed ratio, helps to overcome the difficulty. In practice, this is difficult to measure. Ideally, salinity should be the sum of all dissolved salts in grams per kilogram of sea water. Two properties which are determined by the amount of salt in the sea are conductivity and osmotic pressure. Particle and dissolved matter do affect light absorption in sea water and this influence is used in most optical applications. properties like viscosity, and light absorbtion) are not significantly affected by salinity. The presence of salts influences most physical properties of sea water like density, compressibility, freezing point, temperature of the density maximum to some degree but does not determine them. 
Sea water contains 3.5% salts, dissolved gasses, organic substances and undissolved particulate matter. The solid phase of water is therefore lighter than the liquid phase, which is a rare property. This leads to a sudden expansion in volume, ie a decrease in density. When freezing, all water molecules form tetrahedrons. The physical property is given first, followed by the temperature in ☌ at which the minimum occurs. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
